Peracetic acid (PAA) is a powerful disinfectant with the potential to revolutionize water treatment. It is environmentally friendly, safer to handle and more cost-effective. Because of these benefits, PAA has rapidly become a preferred choice sanitizer, disinfectant, sterilant and bleach for a host of applications across a wide variety of industries. In this article, we will provide a deep-dive into what makes Peracetic Acid the smart choice – not just a viable alternative.
An Effective and Sustainable Solution
The chlorine compounds that industries have relied on for decades to accomplish disinfection and sanitation are wrought with significant drawbacks such as toxicity, corrosiveness, environmental harm and safety hazards. Chlorine is widely used because it is inexpensive and potent however, chlorine has the potential for forming carcinogenic and mutagenic disinfection by-products1 (DBPs).
Peracetic Acid is an eco-friendly alternative that addresses these concerns while providing equal or superior results. PAA boasts a unique chemistry that effectively targets bacteria and other contaminants, has no known DBP formations, rapidly reacts with pollutants, and breaks down into safe chemicals (acetic acid, oxygen and water).
Peracetic acid is an equilibrium mixture of acetic acid and hydrogen peroxide and water. PAA has the chemical formula CH3CO3H and is produced as an equilibrium solution as shown in Figure 1. Commercially available PAA such as the Terrastat® line of products from Brainerd Chemical Company, typically contains a stabilizer to increase storage life.
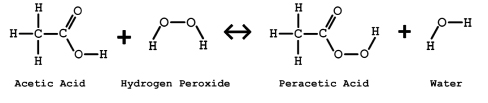
Commercial preparation includes reacting acetic acid with hydrogen peroxide in the presence of a catalyst; specific grades of PAA are formulated by controlling the concentration and amounts of reagents during the manufacturing process.
With a half-life in the order of minutes, PAA typically degrades before entering waterways. The result is an effective disinfection, sanitation, and sterilization agent with minimal potential threat to aquatic environments or reduced water quality. Bioaccumulation in aquatic organisms or in sediments is highly unlikely because PAA has low octanol water partition coefficients (KOW) (0.3, 0.4, and 0.68, respectively) and low sediment adsorption coefficients2 and, PAA typically degrades before entering waterways because its half-life is in the order of minutes.
Extensive testing has shown that this powerful oxidant successfully passes fish toxicity testing and poses no harm to aquatic life. A study conducted by Davidson et al. (2019)3 discovered that PAA does not have any adverse effects on water quality or the performance metrics of rainbow trout, including growth, survival, and feed conversion ratio. To ensure the protection of aquatic ecosystems, the EPA recommends residual PAA concentration at the effluent discharge be below 1mg/1 which is commonly achieved when recommended application guidelines are followed.
Versatility That Goes Beyond Expectations
One of the biggest advantages of PAA is its versatility as a highly adaptable oxidizing that is effective in the order of minutes, is easy handle safely, offers a one-year shelf life when stored properly, it leaves no harmful residues and decomposes into harmless byproducts. In most applications, versus other alternatives, capital cost savings are immediate because retrofitting does not require additional equipment5.
Brainerd Chemical Company stands at the forefront as one of the leading producers of peracetic acid, offering a diverse range of top-tier products and comprehensive services from experienced professionals. With various peracetic acid concentrations, the Terrastat® line of products find extensive utility in chemical synthesis, bleaching, sanitization, disinfection, hygiene, and sterilization across numerous industries.
Safety Doesn’t Have to Be Compromised
Organizations often choose Peracetic Acid because it is equally or more effective without compromising safety.
Traditional disinfectants like chlorine compounds raise concerns about safety hazards to people and our environment. These corrosive substances can cause skin irritation, respiratory problems, and other health issues in people as well as disinfection by-products that wreak havoc on eco-systems. However, Peracetic Acid (PAA) offers a safer, more secure, and environmentally friendly alternative throughout the entire chemical manufacturing process, from production and service to storage, handling, transportation, and disposal.
What sets PAA apart is its safety approvals from regulatory bodies such as the EPA and FDA. With low toxicity levels, PAA is safe for both workers and food products. Its use doesn’t leave any harmful residues, making it environmentally friendly.
When utilized for antimicrobial applications, PAA is typically diluted5 (ranging from 50 to 2,000 ppm in solution). It’s important to note that exposure to PAA in its diluted form as a vapor or mist may cause temporary eye, nose, throat, or respiratory irritation, but these symptoms usually subside once exposure ceases. To minimize any potential over-exposure, it is crucial to implement adequate containment, ventilation, and process controls, thereby reducing direct contact between workers and the vapor, mist, or droplets.
Water Management, Water and Wastewater Treatment
PAA is a game-changer for organizations seeking to mitigate bacterial and effluent wastewater impacts. Facilities are adopting PAA at an exponential rate because it provides more than just an eco-friendly and effective solution – PAA reduces operational headaches and minimizes compliance considerations.
As described in the Alternative Disinfection Methods Fact Sheet: Peracetic Acid’, the need for an economical and relatively simple retrofit to an existing wastewater treatment facility is obvious because of concerns of formation of halogenated disinfection byproducts (DBPs). Peracetic acid (PAA) is a viable alternative to halogenated disinfection chemicals (such as chlorine-based products).
Facilities choose Peracetic Acid because:
- PAA is an effective disinfection compound that does not generate harmful DBPs; toxic residuals and mutagenic or carcinogenic compounds are absent after disinfection.
- PAA is a more rapid acting disinfectant than chlorine-based disinfectants.
- PAA can be economically retrofitted and/or work in series with an existing disinfection system.
- PAA dissipates rapidly and does not generate harmful disinfectant by-products even if overdosed.
- PAA allows for safer and easier handling, storage, and shipping.
- Does not require expensive capital investment to implement.
- Does not require quenching (i.e., no dechlorination).
- Eliminates the possibility of over feeding or under feeding Sodium Bisulfite making it easier to meet regulated discharge permits.
- Normally does not require a Risk Management Plan (RMP).
- Absence of persistent toxic or mutagenic residuals or by-products.
- Small dependence on pH.
- Requires short contact time (minutes vs hours).
- Effective for primary and secondary effluents.
- Effective under varying influent conditions.
PAA is federally approved for use in wastewater disinfection and is currently permitted in approximately 18 states, with several more states in the process of gaining approval. PAA is regulated under the Federal Insecticide, Fungicide and Rodenticide ACT (FIFRA, 7 U.S.C. Section 136)8. North Carolina, New Jersey, Tennessee, West Virginia, Oklahoma, and Texas all have current policies/permitting requirements for PAA usage.
PAA is more effective than Sodium Hypochlorite in wastewater management. It is a stronger oxidizer with oxidation potential higher than that of chlorine or chlorine dioxide9. Significantly shorter contact times (minutes vs hours) are required while using less product to accomplish the same job. And PAA is effective where UV treatment is ineffective in treating industrial wastewater.
Disinfection of wastewater effluents with peracetic acid has been shown to reduce levels of fecal contamination by 97%. The process of disinfection with peracetic acid is easier to manage than other more common methods and the tests performed confirm that from the bacteriological point of view good results can be obtained for urban effluents15.
PAA is proven effective and efficient in treating wastewater at scale for over a decade. The first full scale commercial application of PAA occurred at the St. Augustine Wastewater Treatment Plant, Florida, in 201212 with other large municipalities following such as the Metropolitan Sewer District of Greater Cincinnati13 and the M.C. Stiles Wastewater Treatment Plant14 in Memphis, TN. Their success is paving the way for similar facilities eager to take advantage of all the benefits Peracetic Acid offers.
Broad Application Potential
Industries benefiting from peracetic acid solutions are not limited to municipal and industrial wastewater treatment facilities. PAA benefits food and beverage processing (think fruit and vegetable processing, poultry processing), pharmaceutical manufacturing, environmental remediation, industrial sanitization as well as oil and gas production.
As a broad-spectrum biocide with fast reaction time that leaves no harmful by-products Peracetic Acid is effectively being used as a disinfectant and sterilant in aquaculture, food, oil and gas water treatment, medical, and pharmaceutical industries. Its potency against viruses and bacteria has earned it a reputation as one of the most versatile disinfectants, and as a result, it has become a preferred choice in these industries.
EPA regulation establishes an exemption from the requirement of a tolerance for residues of peroxyacetic acid6, in or on all raw and processed food commodities when used in sanitizing solutions containing a diluted end-use concentration of peroxyacetic acid up to 500 ppm, and applied to tableware, utensils, dishes, pipelines, tanks, vats, fillers, evaporators, pasteurizers, aseptic equipment, milking equipment, and other food processing equipment in food handling establishments including, but not limited to dairies, dairy barns, restaurants, food service operations, breweries, wineries, and beverage and food processing plants. This regulation eliminates the need to establish a maximum permissible level for residues of peroxyacetic acid.
Safer Water Treatment for People and Our Planet
Peracetic acid is recognized as a potent force multiplier that facilitates thorough water treatment and supports a more sustainable future. Compared to traditional chemical treatments, PAA is safe, effective and offers low corrosive qualities while being easy to handle, store, and apply. PAA is a fast-acting disinfectant7. It offers effectiveness, low toxicity, and high biodegradability. This makes PAA versatile delivering powerful performance, minimizing environmental damage, ensuring safety, and reducing the overall cost.
Peracetic acid is making remarkable waves, giving water treatment facilities sustainable and efficient cleaning methods that cater to many different industries. Its versatility, eco-friendliness, and thoroughness have made it the preferred choice for a growing number of plant & facility managers, food, oil and gas producers, and municipalities. By accomplishing tasks faster and reducing operating costs, PAA empowers organizations to achieve more while promoting environmental sustainability that safeguards the planet for all of us.
ABOUT BRAINERD CHEMICAL COMPANY:
Brainerd Chemical Companies dedicated team of engineers, chemists and skilled technicians proudly serve as an industry leader in the manufacturing and supply of peracetic acid. Our expertise in oxygenation and oxidative chemistry sets us apart, enabling us to deliver exceptional solutions through an extensive range of peracetic acid products registered as sanitizers, disinfectants, and antimicrobial agents through the EPA, FDA, and USDA.
Brainerd Chemical Company offers specialty PAA blends under the following tradenames: Terrastat® 15, Terrastat® 15-S, Terrastat® 15-OF, Terrastat® 5-S, Terrastat® 22 and Terrastat® 22-S.
Brainerd Chemical safely manufactures, blends, packages, and distributes custom industrial chemical products and services.
Media Inquiries
1+ 918.622.1214
SOURCE: Brainerd Chemical Company
Citations
- Goveas et al. 2010. Bench-Scale Evaluation of Peracetic Acid and Twin Oxide™ as Disinfectants in Drinking Water, US EPA
- Bell, Kati and Wylie, Varsha. 2016. The Age of Peracetic Acid –A Solution to Increasingly Challenging Regulations, Water Online.
- Davidson et al. 2019. Evaluating the effects of prolonged peracetic acid dosing on water quality and rainbow trout Oncorhynchus performance in recirculation aquaculture systems, Aquacultural Engineering.
- Pileggi et al. 2022. Side-Stream Comparison of Peracetic Acid and Chlorine as Hypochlorite for Disinfection of Municipal Wastewater Effluent at a Full-Scale Treatment Facility, Ontario, Canada. ACS EST Engg.
- Health Hazard Information Sheet Peroxyacetic Acid (PAA), FSIS ESHG-Health-03.00
- U.S. EPA 2000. Peroxyacetic Acid; Exemption From the Requirement of a Tolerance
- U.S. EPA 2012. Alternative Disinfection Methods Fact Sheet: Peracetic Acid
- FIFRA, 7 U.S.C. Section 136
- USDA 2020. Peracetic Acid Processing.
- Bai et al. 2023. Enhanced inactivation of Escherichia coli by ultrasound combined with peracetic acid during water disinfection, Chemosphere.
- Stampi et al. 2001. Evaluation of the efficiency of peracetic acid in the disinfection of sewage effluents, Journal of Applied Microbiology.
- Graham and Skipp. 2015 Peracetic acid replaces chlorine at Florida wastewater treatment plant, The Municipal.
- Garg et al. 2019. Wastewater Disinfection with Peracetic Acid (PAA) and UV Combination: A Pilot Study at Muddy Creek Plant [Cincinnati, OH], EPA/600/R19/084
- Bettenhausen. 2020. How peracetic acid is changing wastewater treatment Chemical and Engineering News.
- Ryther and Champion. 2013. Peracetic acid gains traction as a multifaceted water management tool.
- Au, K and Dunkley, Barry. 2016 Peracetic Acid to Replace Gaseous Chlorine for Wastewater Disinfection: From Bench Scale Testing to Pilot Demonstration. Water Environment Federation pg. 319-330.
- FIFRA, 7 U.S.C. Section 136
- NSF Approval Letter Peracetic Acid 15%
- OSHA Chemical Database PAA
- NIH compound summary
- The Water Resource Foundation. 2019. Application of Peracetic Acid for Municipal Wastewater Processes (PROJECT NO.LIFT14T16/4805)
- Water Environment Federation. 2020. Peracetic Acid Disinfection: Implementation Considerations for Water Resource Recovery Facilities, Access Water.

